How To Get Started With Your Clinical Trials In Australia – Regulatory And Site Perspectives

Australia has the world’s best researchers and health professionals, a world-class research infrastructure, and high standards for clinical trial conduct, which leads to quality, increased speed, and high confidence in the results.
Note: This article was last updated in Oct 2024.

They have quality research facilities, an efficient ethical and regulatory framework, a diverse participant recruitment pool, research tax incentives, and flexibility in conducting research.
Top 7 reasons to conduct clinical trials in Australia
There are several reasons why Australia is an attractive location to conduct clinical trials:
- The fastest route to start clinical trials: Australia offers one of the fastest and most efficient regulatory paths to initiate clinical trials, particularly through the CTN scheme.
- Highly Skilled Workforce: Australia has a highly skilled and educated workforce with a strong focus on medical research. The country boasts of world-renowned researchers and clinicians, making it a hub for innovative medical research.
- Robust Regulatory Framework: Australia has a robust regulatory framework for clinical trials, ensuring the safety of trial participants and the integrity of trial data. The Therapeutic Goods Administration (TGA) is responsible for regulating therapeutic goods, including medical devices and drugs.
- Well-established Infrastructure: Australia has well-established infrastructure and facilities for clinical trials. This includes state-of-the-art hospitals, research institutions, and clinical research organizations (CROs) that can provide high-quality services and support for clinical trials.
- Access to Patient Populations: Australia has a diverse patient population, making it an ideal location for clinical trials that require a range of patient demographics. Additionally, Australia has a universal healthcare system, which means that patients have access to healthcare services regardless of their ability to pay.
- Competitive Cost Structure: The cost of clinical trials in Australia is competitive with other developed countries, like the U.S. and Western Europe, and the government provides financial incentives for research and development.
- Favorable Time Zone: Australia’s time zone is advantageous for clinical trials collaborating with the Asia-Pacific region, facilitating work with key pharmaceutical players like China, Japan, and South Korea.
Click here to learn more about why you should conduct your clinical trials in Australia.
Along with these reasons, questions may arise for those interested in Australia for conducting clinical trials, which include
- What are the regulatory processes (hurdles?), and
- How can we find the sites to conduct clinical trials there?
In these articles, let’s try to address these questions.
Regulatory for Clinical Trials in Australia
The Therapeutic Goods Administration (TGA) is the regulatory body in Australia. The TGA regulates the quality, supply, and advertising of medicines, pathology devices, medical devices, blood products, and many other therapeutics.
Australian sponsor
- In order to conduct clinical trials in Australia, you generally need an Australian sponsor or a local representative.
- The Therapeutic Goods Administration (TGA), which regulates clinical trials in Australia, requires that the sponsor of a clinical trial must be an Australian entity or have a local representative if the sponsor is based overseas.
The sponsor may be an,
- Individuals (medical practitioners),
- A body or organization (hospitals, area health services, non-government organizations), or
- A company (pharmaceutical companies, CROs).
TGA would deal directly with the Australian sponsor on all matters relating to the trial.
Human Research Ethics Review (HREC)
- Clinical trials must be approved by an HREC, which ensures the trial is ethically sound and protects participant rights.
- Submitting the trial to an accredited HREC for approval is often the first step before engaging with the TGA.
Ethics committees in Australia
- The ethics committee must have notified its existence to the Australian Health Ethics Committee (AHEC) of the National Health and Medical Research Council (NHMRC) and provided assurances that it is operating within its guidelines.
- Ethics committees in Australia provide a combined ethical and scientific review process,
- HRECs have a pivotal role in clinical research in Australia because they undertake the key responsibilities of clinical trial approval and oversight
- HRECs operate under the auspices of the Australian Health Ethics Committee.
- Additionally, public hospitals must undertake a research governance step by conducting a site-specific assessment (SSA) for each project.
- Therefore, public sites must obtain both HREC and SSA approval for a clinical trial to commence.
- The institution or organization conducting the trial will give the final go-ahead, taking into account the advice provided by the HREC.
TGA
- CTN research projects must not start at a site until the research project and investigational site have been notified to the TGA and the appropriate notification fee paid.
- It is preferable to wait for the TGA to confirm receipt of the CTN before recruitment commences.
Research Governance Office (RGO) approvals
- According to AUS-43 and the G-TrialsSOP, all participating institutions must also authorize any research in accordance with their research governance frameworks, including site-specific assessments (SSAs) for public health institutions. The SSA and ethics review (incorporating scientific review) may occur in parallel.
- If the sponsor chooses private sites to conduct clinical trials may not require RGO approval. if the sponsor chooses to include public sites will require RGO approval.
Application process for clinical trials in Australia
In Australia, the clinical trial approval process involves two primary pathways for obtaining regulatory and ethical approval:
- Clinical Trial Notification (CTN) scheme and
- Clinical Trial Exemption (CTX) scheme.
Clinical Trial Notification (CTN) Scheme
The CTN Scheme is the most commonly used regulatory process for clinical trials in Australia. It is appropriate for trials involving investigational products (including drugs, biologics, and medical devices) that do not require prior approval from the Therapeutic Goods Administration (TGA). Instead, the sponsor notifies the TGA about the trial and provides details of the investigational product.
Steps in the Clinical Trial Notification (CTN) scheme
- Ethics Committee Approval: A Human Research Ethics Committee (HREC) must first review and approve the clinical trial.
- Institutional Approval: The institution conducting the trial must also grant approval.
- Notification to the TGA: After obtaining the necessary approvals, the trial sponsor submits a notification to the TGA that includes trial details and information about the investigational product.
Key points
- The TGA does not review the trial in detail but must be notified.
- Approval and oversight responsibilities rest mainly with the HREC and the institution.
- The scientific validity of the trial design
- The risks and benefits of the therapeutic product
- The acceptability of the trial procedure from an ethical standpoint
Approval time in CTN Scheme
- The total approval process under the CTN Scheme can range from 6 to 12 weeks, depending on the speed of the HREC and institutional reviews.
- Since the CTN Scheme does not require the TGA to review the trial in detail, the timeline primarily depends on how quickly the ethics and institutional approvals are completed.
- Once the ethics committee and institutional approvals are obtained, the trial sponsor immediately notifies the Therapeutic Goods Administration (TGA).
- After the sponsor submits the notification, the TGA acknowledges it formally within 1–2 business days.
Clinical Trial Exemption (CTX) Scheme
The CTX Scheme is used for higher-risk trials, where the investigational product may pose greater risks to participants. This process requires the TGA to review the scientific, safety, and ethical aspects of the trial before it can begin.
Steps in the Clinical Trial Exemption (CTX) scheme
- TGA Review: The sponsor submits an application to the TGA for review. The TGA assesses the safety of the investigational product and the trial design.
- Ethics Committee Approval: Once the TGA grants approval, an HREC must review and approve the trial.
- Institutional Approval: The institution conducting the trial must also grant approval.
Key points
- The TGA conducts a thorough assessment of the trial before approval.
- Required for trials involving high-risk or novel products that may have insufficient safety data.
Clinical Trial Approval Scheme (CTA) – forms
There are two forms, each reflecting separate processes (Parts), that must be submitted to TGA by the sponsor.
- Part 1 constitutes the formal CTA application. The sponsor of the trial must complete it and submit it to the TGA with data for evaluation.
- Part 2 notifies the commencement of each new trial conducted under the CTA, as well as new sites in ongoing CTA trials. The sponsor must submit the Part 2 form within 28 days of commencing the supply of goods under the CTA.
Approval time in CTX Scheme
- The approval timeline for the Clinical Trial Exemption (CTX) Scheme in Australia is typically longer than the CTN process due to the additional regulatory review by the Therapeutic Goods Administration (TGA).
- The TGA review process usually takes around 30 to 50 working days (approximately 6 to 10 weeks). The TGA conducts a detailed assessment of the investigational product, including its safety data, manufacturing information, and trial design.
- Following TGA approval, the HREC review typically takes 4–8 weeks.
- Site-specific institutional approval generally takes another 2–4 weeks, similar to the CTN process.
- The overall approval timeline for the CTX scheme is typically 3 to 6 months. This includes:
- TGA review: 6–10 weeks.
- HREC and institutional approvals: 6–12 weeks combined.
The approval process flow for clinical trials in Australia
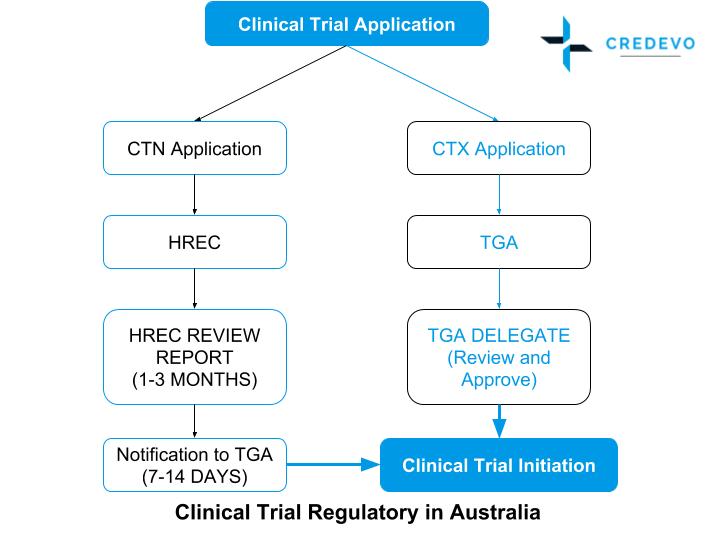
Legends: CTN– Clinical trial Notification, CTX– Clinical trial Exemption, TGA-Therapeutic Goods Administration, HREC-Human Research Ethics Committee
NOTE: HREC Review time depends upon the institution
Fee structure for clinical trials in Australia
Here are the fees/charges for various categories.
| Category | Fee |
|---|---|
| Prescription medicine, biological, and other listed and registered therapeutic goods CTN. | $345 AUD |
| Prescription medicine CTX for 30-day evaluation. | $1,665 AUD |
| Prescription medicine CTX for 50-day evaluation | 20,800 AUD |
| Biological CTX | $25,200 AUD |
- Applications for clinical trials under the CTX scheme cost more than notifications under the CTN scheme because of the increased work needed to evaluate the CTX application data.
- Applications for clinical trials under the CTX scheme cost more than notifications under the CTN.
One can find more information about them on the TGA website.
Medical device trials
The clinical trial regulatory process for devices is essentially similar to that for medicines. Often, the protocol is sent in by the sponsor/principal investigator(s) with the CTN notification form, and in such instances, comments are provided on the document by the medical advisory staff. CTX applications for medical devices are rare.
Top benefits of conducting clinical trials in Australia
1. Quality research and facilities
for its highly trained clinical workforce and the high-quality data produced by its experienced, trained (and accredited) research teams.
2. Efficient ethics and regulatory framework
- Research proposals are submitted directly to Australian Human Research Ethics Committees (HRECs) which assume the primary review responsibility for ethical and scientific review.
- The usual review cycle takes only 4 to 8 weeks and is based on the submission of a protocol, investigator brochure, and if required, an independent toxicology report.
3. The diverse participant recruitment pool
Australia has an informed and willing population of potential trial participants, resulting in a higher level of research participation than would be expected from a population of its size.
4. Investment Incentives
- The Australian Government’s generous Research & Development (R&D) Tax Incentive encourages more industry investment in R&D.
- The Incentive provides businesses investing in eligible R&D with generous tax offsets:
- A 45 percent refundable R&D tax offset (equivalent to 45c per $1 of eligible R&D) for companies with an aggregated annual turnover of less than A$20 million; and
- A 40 percent non-refundable R&D tax offset (equivalent to 40c per $1 of eligible R&D) for all other eligible companies.
5. Flexibility
- Companies conducting clinical trials in Australia do not require the US FDA Investigational New Drug (IND) application approval.
- Data output from studies carried out in Australia meets global standards and can be used to support international regulatory applications, including the US FDA IND submission. This makes the process more efficient, flexible, and ethical.
- Australia offers a lot to all kinds of drug / medical device/biotech companies in terms of market expansion. It’s always interesting to consider Australia in your global strategy.
Need Support in Australia or Have questions?
We’d love to help you conduct clinical trials in Australia. Provide your details below to connect with us and explore our expert services in Australia.