Pancreatitis – Global Clinical Trial Feasibility

Pancreatitis, inflammation of the pancreas, is the leading cause of GI-related hospitalizations in the USA and many other countries, with a medical condition requiring emergent care.

The yearly incidence of Acute Pancreatitis in the United States and western countries is approximately 70-80 new cases per 100,000 population and has increased over the last decade. In 2015, globally, about 8.9 million cases of pancreatitis occurred, which resulted in 132,700 deaths.
What Is Pancreatitis?
Acute pancreatitis is inflammation of the pancreas, usually accompanied by abdominal pain known as pancreatitis pain and elevations of serum pancreatic enzymes. acute pancreatitis may lead to chronic pancreatitis.
In chronic pancreatitis, inflammation has resolved, but with resultant damage to the gland, is characterized by fibrosis, calcification, and ductal inflammation.
Causes of Acute Pancreatitis
The most common causes of acute pancreatitis are gallstones, heavy alcohol use followed by viral, malignancy, metabolic, ischemia, direct trauma, smoking, and certain medications.
Recently, Avicii, a Swedish musician, Dj, remixer, died on 20 April 2018, at an age of 28, who had previously suffered from pancreatitis. Avicii’s excessive drinking was thought to be caused to his health condition and death.
Some of the drugs well-recognized to cause pancreatitis include Azathioprine, 6-Mercaptopurine, Trimethoprim-Sulfamethoxazole, Pentamidine, Asparaginase, Methyldopa.
How is Pancreatitis classified?
Mild, moderate, or severe based on physiological findings, laboratory values, and radiological imaging.
Pancreatitis may be further classified into
- Acute interstitial and
- Acute hemorrhagic disease.
In acute interstitial, the gland architecture is preserved but is edematous. Inflammatory cells and interstitial edema are prominent within the parenchyma.
Hemorrhagic disease is characterized by marked necrosis, hemorrhage of the tissue, and fat necrosis. There is marked pancreatic necrosis along with vascular inflammation and thrombosis.
Prevalence of Pancreatitis
- Eighty percent of acute pancreatitis cases in the United States are related to alcohol use or biliary stones.
- Globally the incidence of acute pancreatitis is 5 to 35 cases per 100,000 people.
- The incidence of acute pancreatitis in the UK ranges from 150 to 420 cases per million population and is currently rising.
- There is considerable geographical variation. The incidence in the UK and the Netherlands is relatively low compared to Scandinavian countries and the USA.
- The incidence of acute pancreatitis has wide variability within populations, with about 1–5 cases per 10,000 population per year.
- Gallstones and alcohol are the most common cause of pancreatitis in the United States and other western countries.
- Biliary tract disease accounts for 35–50% of all cases. (2)(5).
Treatment of Pancreatitis
Pancreatitis treatment depends on its type.
There are no established targeted pharmacologic therapies for Pancreatitis. Most of the treatment is through management. New research is being conducted to find better treatment/management therapies.
- For moderately acute pancreatitis, it includes fluid, opioids (to relieve pain), oxygen (occasionally).
- In the case of severe acute pancreatitis, it involves admission ICU, monitoring of heart function, vital signs and urine production, fluids, oxygen, nutrients through feeding tubes, and ERCP procedure (in case of gallstones).
- Recent advancements in pancreatitis management include early aggressive fluid resuscitation and isotonic crystalloid fluids.
- No current targeted pharmacologic therapies are recommended or available to treat acute pancreatitis. (2)(3)
- New research is being conducted worldwide to bring out therapies to treat (or, manage) Pancreatitis. Due to the impact on the quality of life, pancreatitis has received widespread attention from researchers worldwide.
- Several such treatments have reached the clinical development stage. Clintrial.gov shows many such clinical trials on pancreatitis worldwide. These are being discussed in the next section.
Clinical Trials on Moderately and Severe Acute Pancreatitis
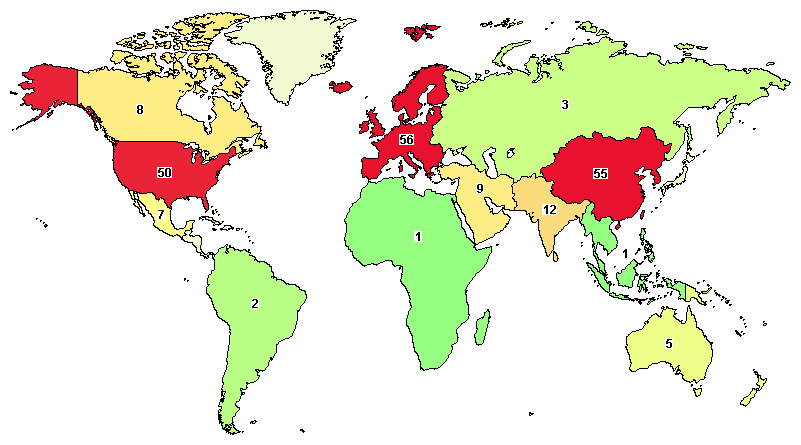
Total clinical trials registered on Acute Pancreatitis going around the globe are 225, where the major clinical studies are going in the United States (50), China (47), India (11), and Spain (10).
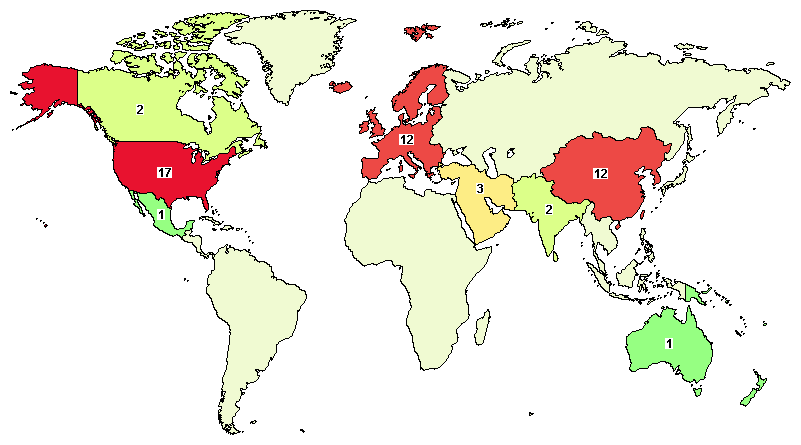
The total on-going studies are 39, with major clinical studies in the United States (17), China (8), United Kingdom (2), and India (2).
[Data as on Sep-2020]
Pancreatitis Global Clinical Trial Feasibility on CREDEVO
A global clinical trial on Pancreatitis is being explored. The feasibility of this trial was conducted on credevo across multiple regions including the USA, India, and Europe.
- This Phase III clinical trial on acute pancreatitis was a randomized, double-blind, placebo-controlled, parallel-group, multi-center study to assess the efficacy and safety of study drug against placebo in study subjects. The study drug was investigated was Ulinastatin at a therapeutic dose.
- The clinical trial was evaluated in medical centers and sites located in South Asia, United States, United Kingdom, and other European countries.
- The primary objective of the trial is to assess the efficacy with safety as a secondary objective.
- Diagnosis of acute pancreatitis must include upper abdominal pain, serum amylase and/or lipase, or characteristic findings on contrast-enhanced CT/ MRI/ultrasonography (USG).
- Males and females, aged between 18 to 70 years were included in this clinical study.
Feasibility Results
Clinical trial feasibility was assessed with sites/investigators from Europe (Denmark, France, Greece, Italy, Norway, Spain, Switzerland, and the United Kingdom), the USA, and India.
Feedback, from sites/investigators with patient recruitment potential, were from countries as follows
| S No. | Country | Participation |
| 1 | Denmark | 20 patients / month |
| 2 | India | 5 patient / month |
| 3 | USA | 4 patients / month |
| 4 | Greece | 1 patient / month |
| 5 | Spain | 1 patient / month |
Contact Investigators From This Feasibility Assessment
Would you like to contact these investigators and assess feasibility with them for your clinical trial?
Now, it’s very easy to do so.
Just provide your email below and further details will be sent to you. Don’t worry, your email will not be published or shared.
For other details, click here to contact us.
References
- https://en.wikipedia.org/wiki/Acute_pancreatitis
- https://en.wikipedia.org/wiki/Pancreatitis#Severe_acute_pancreatitis
- https://www.msdmanuals.com/home/digestive-disorders/pancreatitis/acute-pancreatitis
- https://www.hopkinsmedicine.org/gastroenterology_hepatology/_pdfs/pancreas_biliary_tract/acute_pancreatitis.pdf
- https://patient.info/doctor/acute-pancreatitis-pro
- https://clinicaltrials.gov/ct2/results/map?cond=Pancreatitis%2C+Acute&map=
- https://www.msn.com/en-us/health/medical/what-is-pancreatitis-why-avicii-stopped-performing/ar-AAw7kvH?ocid=spartandhp
- https://clinicaltrials.gov/ct2/results/map?recrs=ad&cond=Pancreatitis%2C+Acute&map=
- https://www.hopkinsmedicine.org/gastroenterology_hepatology/diseases_conditions/faqs/chronic_pancreatitis.html
- http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.831.8589&rep=rep1&type=pdf