Critical Factors In Selecting A CRO: From Frost & Sullivan White Paper

Outsourcing in the clinical trial industry has been on the rise for the last several decades. This has been due to a combination of several factors, including the increasing cost of research, blockbuster molecules going off-patent causing revenue loss, and lack of extensive in-house R&D infrastructure. This has resulted in an interesting situation, where increasingly more clinical trials are being outsourced to contract research organizations (CROs) worldwide.

The latest white paper from Frost & Sullivan discusses a lot about the clinical research industry, including a comparison of clinical trials conducted in Asia and the west, and various factors suggested to be critical for selecting CROs in Asia.
In our earlier article on Potential and Challenges with Clinical Trial Sites in Asia & West, we had discussed clinical sites in Asia & West on the basis of this white paper. In this article, let’s discuss the critical factors suggested in this white paper for selecting CROs.
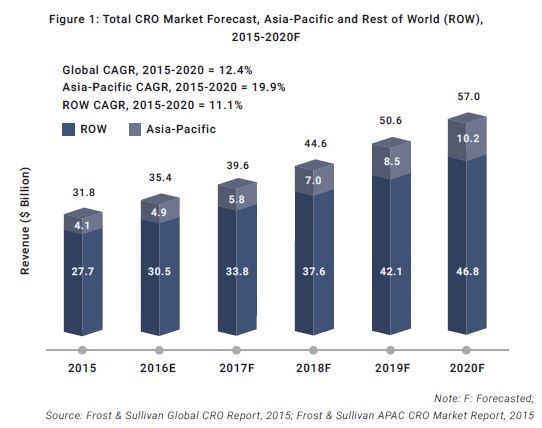
Most of these factors were related to three primary considerations, namely Capability, Experience, and Network & Partnerships. While considering a particular CRO, there are specific questions recommended to be asked for each of these factors before selection.
Critical factors
Capability
Sponsors would need a CRO having capabilities that can ensure knowledgeable teams, client focus, and the ability to provide customized solutions. It’ll help to know
- Size of team
- Training given to the team
- Technology at disposal, Infrastructure and IT systems
- Integration in services across clinical trial value chain
- Culture for customer service
- Regulatory know-how for local as well as US & EU markets
- Communication styles, including languages spoken by team (English?)
Experience
It’s of great importance to know about the experience of CRO as well. The extent, breadth, and quality of CROs teams’ experience can significantly affect the value of their service. It helps to ask the following questions.
- How many projects were performed?
- Which regions has CRO teams worked for?
- Study types, phases, diseases and therapeutic areas?
- Are there case studies of CROs’ performance, e.g. rescue studies?
- How many countries does the CRO have the presence in?
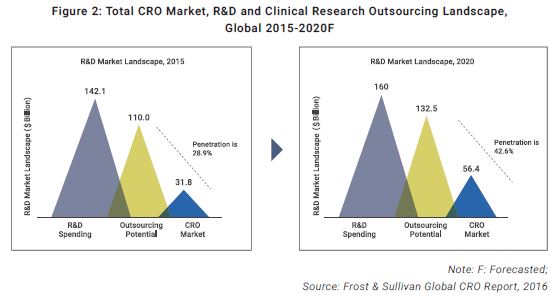
Network & Partnerships
A CRO can tremendously enhance the value of their core capability and experience by having a useful networks and partnerships.
- Besides their own presence, does CRO has a working partnership with other companies to complement their strengths and reach?
- Which countries can be additionally covered with these partnerships?
- Which investigators, KOLs and experts are included in their network?
- Does CRO have an affiliation with any physician association?
- Can CRO work with systems and SOPs of other organizations that they have a partnership with?
- Does CRO have a referral from partners that they have worked with earlier?
Cost
While, the white paper explains the above factors considering the Asian CROs, which traditionally have lower charges than western CROs, it is extremely important to consider cost in a global context.
With other factors being comparable, trial management may find it lucrative to go for CRO with the best price offer. While prices differ greatly across geographies, prices may differ across the CROs within the same region too, and it always helps to compare them.
Click here to know How To Save Upto 80% Of Cost And Time In Initiating Clinical Trials
What was your experience?
Have you followed these factors or other ones, while selecting a CRO?
Or, if you work at a CRO, how does it fare on these factors?
Provide your comments below or send them to [email protected].
One thought on “Critical Factors In Selecting A CRO: From Frost & Sullivan White Paper”
Comments are closed.