Amyotrophic Lateral Sclerosis (ALS): Current Treatment Landscape & Emerging Opportunities
Key Takeaways from this article
— Amyotrophic Lateral Sclerosis (ALS) affects approximately 30,000 Americans at any given time, with about 5,000 new diagnoses each year in the US alone.
— The global ALS treatment market is in an active growth phase, driven by genetic stratification, biomarker-enabled trials, and a first genotype-specific approval in 2023.
— C9orf72 ALS, the largest genetic subgroup, remains without an approved therapy, representing the most significant near-term first-mover opportunity.
— TDP-43 pathology, present in the vast majority of ALS cases, represents the broadest commercial target in the pipeline.
— Regulatory infrastructure, trial networks, and patient advocacy ecosystems are more mature in ALS than in almost any other rare neurological disease.
— Presymptomatic intervention trials are redefining the addressable population for the next generation of disease-modifying therapies.
Amyotrophic Lateral Sclerosis (ALS) is one of the most devastating neurological diseases known to medicine, universally fatal, relentlessly progressive, and historically resistant to therapeutic intervention. Yet the landscape is changing. The approval of the first genotype-specific therapy in 2023 marked a turning point. Clinicians and researchers now treat ALS not as a single, monolithic disease but as a syndrome of molecularly distinct subtypes, each with its own therapeutic logic.

This article provides an overview of the ALS treatment landscape, including clinical realities, standard of care, pipeline developments, regulatory environment, and strategic opportunities. It is intended to inform decision-making across R&D, regulatory, and commercial functions.
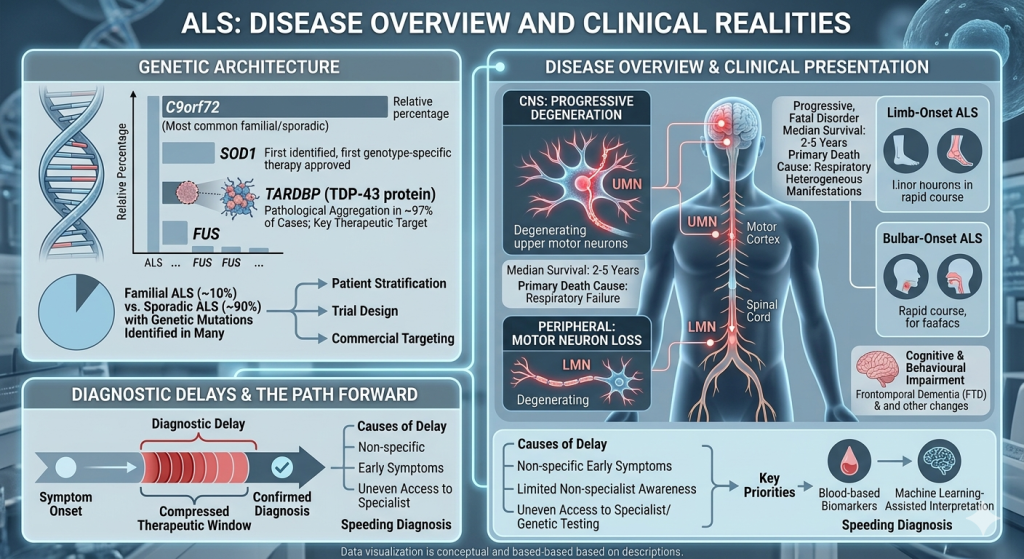
1. Overview and Clinical Realities of Amyotrophic Lateral Sclerosis (ALS)
ALS progressively degenerates upper motor neurons in the motor cortex and lower motor neurons in the brainstem and spinal cord. Median survival from diagnosis is two to five years, with respiratory failure as the primary cause of death.
Clinical Presentation of Amyotrophic Lateral Sclerosis (ALS)
The disease manifests heterogeneously. Limb-onset ALS, the most common form, accounts for the majority of cases and typically presents with progressive weakness and atrophy in the arms or legs. Bulbar-onset ALS presents with speech and swallowing difficulties and generally carries a more rapid course. Cognitive and behavioural impairments are more common than previously recognized, with frontotemporal dementia in some patients and broader cognitive changes in many more.
Genetic Architecture
More than 40 genes have been implicated in ALS. The four most clinically significant are C9orf72, SOD1, TARDBP, and FUS. C9orf72 hexanucleotide repeat expansion is the most common genetic cause, accounting for a substantial proportion of both familial and sporadic cases. SOD1 was the first ALS gene identified and is now the target of the only approved genotype-specific therapy. TTARDBP encodes TDP-43, whose pathological aggregation occurs in most ALS cases, making it the most broadly relevant therapeutic target.
Approximately 10% of cases are familial, but researchers identify underlying genetic mutations in a significant proportion of apparently sporadic cases, directly impacting patient stratification, trial design, and commercial targeting.
Diagnostic Delays and Their Consequences
Average time from symptom onset to confirmed diagnosis remains significant, compressing the therapeutic window and limiting eligibility for early-stage trials. Delays stem from non-specific early symptoms, limited awareness, and uneven access to specialists. Improving diagnosis with biomarkers and AI-assisted interpretation is a key priority.
2. Current Treatment Landscape of Amyotrophic Lateral Sclerosis (ALS)
ALS management combines approved disease-modifying pharmacotherapy with comprehensive multidisciplinary supportive care. Until recently, approved options offered only modest symptomatic benefit. The 2023 approval of tofersen fundamentally changed the trajectory of the field.
Approved Therapies
- Riluzole: First ALS therapy, modest survival benefit, now generic.
- Edaravone: Slows functional decline in early, rapidly progressing patients.
- Tofersen: First genotype-specific therapy targeting SOD1, FDA/EMA approved, sets template for future programs.
Supportive and Multidisciplinary Care for Amyotrophic Lateral Sclerosis (ALS)
Beyond pharmacotherapy, optimal ALS management requires coordinated input from neurology, respiratory medicine, nutrition, speech therapy, physiotherapy, and mental health services. Non-invasive ventilation remains the most impactful life-prolonging intervention after pharmacotherapy and is a critical component of comprehensive care in specialist ALS centres.
Where Current Therapies Fall Short
Despite these advances, significant limitations persist. No approved therapy addresses the neurological and cognitive dimensions of the disease in neuronopathic patients. C9orf72 ALS, the largest genetic subgroup, remains entirely without a targeted treatment. And TDP-43 pathology, which underlies the overwhelming majority of all ALS cases, has no approved therapeutic intervention. These gaps directly shape the pipeline priorities described in the following section.
3. The Evolving Pipeline for Amyotrophic Lateral Sclerosis (ALS): From Symptomatic to Disease-Modifying
The ALS pipeline is highly active, with companies pursuing diverse approaches, evolving from small-molecule symptom treatments to precision genetics, novel biologics, and early interventions.
Gene-Targeted Therapies
The Tofersen approval has catalysed a wave of genotype-specific programmes. Antisense oligonucleotide approaches targeting C9orf72 are in active clinical development, addressing the largest unmet genetic subset. FUS-ALS, associated with particularly aggressive early-onset disease, is also the subject of an advanced clinical programme. AAV-based gene therapy offering potential one-time dosing is being explored for SOD1-ALS, with implications for long-term adherence and commercial differentiation.
TDP-43 Directed Approaches
Given the near-universal presence of TDP-43 pathology in ALS, this represents the largest potential commercial target in the pipeline. Approaches under development include restoration of downstream targets disrupted by TDP-43 dysfunction, as well as direct targeting of TDP-43 aggregation itself through novel biological delivery mechanisms. Early clinical data are anticipated in the near term and will be closely watched by the field.
Immunomodulatory and Small-Molecule Approaches
A parallel track of development focuses on neuroinflammation and secondary disease pathways. Regulatory T-cell modulation has generated significant commercial interest, with one programme reaching a major licensing transaction. Small-molecule approaches — including fixed-dose combination strategies — have demonstrated encouraging signals in Phase 2 and are advancing toward confirmatory studies. First-in-class mitochondrial pathway inhibitors have recently entered Phase 1, adding further mechanistic diversity to the pipeline.
Presymptomatic Intervention
Perhaps the most strategically significant frontier is intervention before symptom onset. Trials evaluating disease prevention in presymptomatic carriers of known ALS mutations are underway, potentially expanding the addressable population to include asymptomatic individuals identified through genetic testing. If successful, this approach would fundamentally redefine the commercial scope of genotype-specific therapies.
4. Clinical Development: Navigating a Complex but Maturing Environment
ALS presents distinct clinical development challenges, but also a more mature trial infrastructure than most rare neurological diseases. Understanding both is essential for any organisation entering or expanding in this space.
Trial Design Challenges
Disease heterogeneity in onset site, progression rate, and genetic architecture complicates endpoint selection and patient stratification. The relatively short survival window compresses trial timelines and creates urgency around recruitment. Standard-of-care variability across regions can affect both baseline comparisons and the interpretation of outcomes.
Enablers: Infrastructure and Biomarkers
Counterbalancing these challenges is an exceptional trial infrastructure. Established clinical networks with experience across dozens of sites enable multinational recruitment at scale. Platform trial designs with shared placebo groups have meaningfully reduced development time and cost. The FDA’s acceptance of neurofilament light chain reduction as a surrogate endpoint reasonably likely to predict clinical benefit has de-risked development programmes and opened accelerated approval pathways for developers.
Real-world data infrastructure — including pooled clinical datasets from thousands of patients across multiple trials and natural history studies — provides unprecedented support for endpoint selection, trial simulation, and regulatory submissions.
5. Regulatory Pathways: Established Frameworks, Evolving Expectations
ALS development benefits from well-established regulatory frameworks that provide both incentives and accelerated pathways for developers. The Tøfersen approval has now created a concrete precedent that subsequent programmes can reference and build upon.
Key Mechanisms Available to Developers
- Orphan Drug Designation: Seven-year US market exclusivity, tax credits, and fee waivers. The ultra-rare nature of genetic ALS subsets ensures consistent eligibility.
- Breakthrough Therapy Designation: Available for therapies addressing serious conditions with preliminary evidence of substantial improvement, meaningfully accelerates development timelines.
- Accelerated Approval: Enables approval based on surrogate endpoints reasonably likely to predict clinical benefit. NfL reduction is now validated in this context following the Tofersen precedent.
- Global Harmonisation: Differences in trial design expectations, endpoint standards, and post-approval requirements across the FDA, EMA, and other agencies require early multi-agency engagement.
The integration of real-world evidence is increasingly expected by regulators, particularly for ultra-rare genetic subsets where large randomised controlled trials are not feasible. Early and proactive regulatory engagement, including pre-IND meetings and parallel scientific advice, is a critical success factor in ALS development programmes.
6. Market Access and Commercial Considerations
The commercial landscape for ALS therapies is characterised by high treatment costs, evolving payer expectations, and significant regional variability in access and reimbursement. Regulatory strategy and market access planning must be aligned from early in development; the type of approval pathway chosen has direct downstream implications for payer evidence requirements.
Pricing and Reimbursement Dynamics
Approved ALS therapies are among the highest-priced in neurology, reflecting the orphan designation, small patient populations, and significant unmet need. However, cost-effectiveness scrutiny is intensifying. Health technology assessment bodies in several markets have raised concerns about the ratio of clinical benefit to cost for existing therapies, creating a challenging environment for premium pricing of future entrants without strong outcome data.
Access Barriers
- Genetic testing coverage: Reimbursement for genetic testing, essential for identifying eligible patients for genotype-specific therapies, remains inconsistent across markets, with some payers classifying it as experimental.
- Specialist centre concentration: Specialist ALS centres with the infrastructure to administer and monitor advanced therapies are concentrated in high-income countries, limiting commercial reach in emerging markets.
- Diagnostic delay: The diagnostic delay from symptom onset to confirmed diagnosis compresses the window during which patients are eligible for early-stage interventions, affecting both trial recruitment and commercial uptake.
Strategic Partnership Opportunities
Patient advocacy organisations play an unusually powerful role in ALS relative to most rare diseases, in funding research, accelerating trial recruitment, driving policy change, and shaping public awareness. Early engagement with advocacy infrastructure is not merely a commercial best practice but a genuine strategic differentiator in this field.
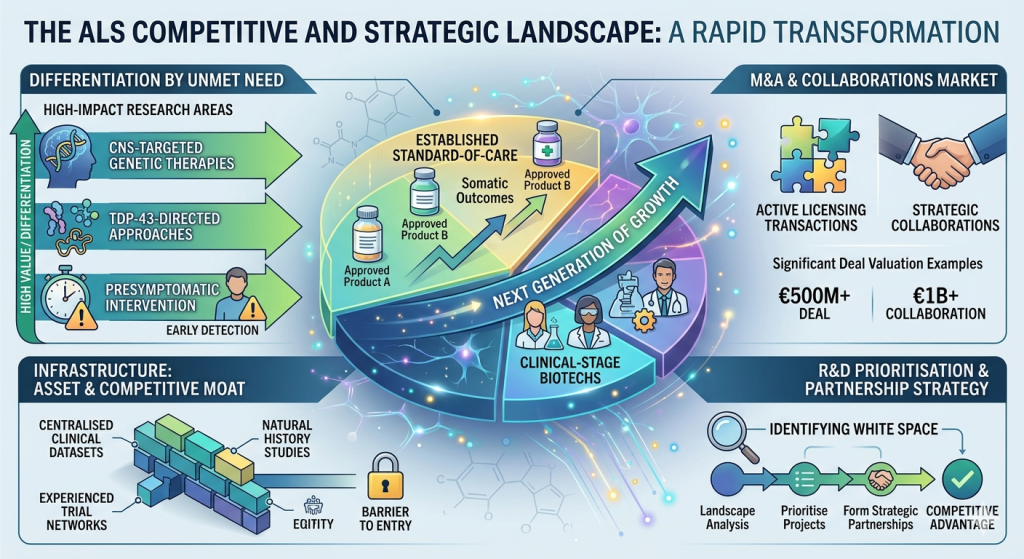
7. Competitive and Strategic Landscape
The ALS competitive landscape is undergoing rapid transformation. Established pharmaceutical companies hold approved products in the current standard-of-care market, but the next generation of growth is being driven by clinical-stage biotechs advancing precision genetic and biological approaches.
Differentiation in this space increasingly depends on addressing the most significant unmet needs, CNS-targeted genetic therapies, TDP-43-directed approaches, and presymptomatic intervention rather than competing on incremental improvements to existing somatic outcomes. Licensing transactions and collaborations are active, with several deals reaching significant valuations as the field matures.
The diagnostic and research infrastructure supporting ALS development, including centralised clinical datasets, natural history studies, and experienced trial networks, provides both an asset and a competitive moat for organisations that engage with it early. Understanding where white space exists within this landscape is essential to R&D prioritisation and partnership strategy.
8. Where the Real Opportunities Lie
Despite meaningful recent progress, ALS remains a disease where the largest opportunities are still ahead. The following areas represent the highest-potential zones for innovation and strategic investment over the coming decade.
C9orf72-Targeted Therapy
The largest genetic subgroup in ALS has no approved therapy. The patient population is well characterised, relevant biomarkers are defined, and the regulatory pathway is established following the tofersen precedent. A first-mover approval in C9orf72-ALS would represent a significant commercial milestone and establish a platform for lifecycle extension.
TDP-43 Pathology: The Broadest Commercial Target
Present in the vast majority of all ALS cases spanning both sporadic and familial forms, TDP-43 pathology represents the ultimate commercial prize. Multiple mechanistic approaches are in development. Early clinical signals will be defining events for the field.
Combination Therapy Platforms
As in oncology, optimal long-term outcomes in ALS will likely require combination approaches targeting multiple disease pathways simultaneously. Intellectual property around rational combination strategies and lifecycle management for approved products represents underappreciated strategic assets.
Early and Presymptomatic Intervention
Biomarker-enabled early diagnosis and trials in presymptomatic genetic carriers are redefining the addressable population. If prevention trials generate positive data, the commercial scope of genotype-specific therapies expands significantly — from treatment of established disease to true disease prevention.
Digital Health and Real-World Evidence
Remote monitoring tools, wearable sensors, and digital biomarkers are increasingly integrated into ALS trials and clinical practice. These technologies not only improve trial efficiency and patient convenience but also generate real-world evidence that supports both regulatory submissions and payer negotiations.
Access the Full Amyotrophic Lateral Sclerosis (ALS) Intelligence Report
For those who found this overview useful and are seeking a more comprehensive foundation for strategic decisions, the full research report is available. It has been developed specifically to support pharmaceutical and biotech professionals navigating this rapidly evolving field — covering areas that this article, by design, introduces rather than exhausts.
We would be glad to assist you in determining whether the complete report is the right resource for your needs. You are warmly welcome to reach out, and the full report is available for those who wish to access it. Access the complete report
9. Future Outlook of Amyotrophic Lateral Sclerosis (ALS)
The therapeutic landscape for ALS is undergoing a fundamental transformation, shifting from symptomatic management toward targeted, disease-modifying, and potentially preventive interventions. The approval of the first genotype-specific therapy has validated the precision medicine model for ALS and opened a regulatory template that subsequent programmes are already following.
The coming decade will be defined by the expansion of genotype-specific approaches to C9orf72 and other genetic subsets, the emergence of first-generation TDP-43-directed therapies, and the development of rational combination regimens targeting multiple disease pathways simultaneously. Advances in blood-based biomarkers and machine learning-assisted diagnosis will progressively compress diagnostic delays, expanding the pool of patients eligible for early intervention.
For patients and families affected by this relentless disease, these developments represent genuine and substantive grounds for optimism. For pharmaceutical and biotech stakeholders, they represent one of the most compelling and well-supported development environments in rare neurological disease, a field where scientific momentum, regulatory clarity, infrastructure maturity, and unmet need have converged at the same moment.
Need Support in Rare Disease Clinical Research?
Rare diseases present a unique set of challenges across the clinical development continuum, from early-stage strategy and study design through to the planning and execution of clinical trials. These challenges are often compounded by limited patient populations, evolving regulatory expectations, and gaps in clinical and real-world data.
With experience supporting organizations working in the rare disease space, our team can contribute across different stages of development, including strategic insights, feasibility assessments, and clinical trial execution.
If you would like to explore specific requirements or discuss potential areas of support, please feel free to share your details through the form below. Our team will review your request and connect with you accordingly.